Abstract
This study aimed to determine the causative agent of black spots which observed in the mesentery, adipose tissue and peritoneum of orange-spotted grouper, Epinephelus coioides. The parasitological examination using light and electron microscope confirmed infection with trichosomoidid parasite, genus Huffmanela, which identified as Huffmanela japonica (H. japonica). The examined eggs were present in two stages (stage 1, mature light brown eggs with larva; stage 2, advanced dark brown eggs with grown larva). The histopathological alteration of the infected tissues together with determination of immunological gene expression in different organs demonstrated prominent response of the host tissues against the studied parasite. Pathological examination of the infected fish revealed that the lesions were mainly in the mesenteric and diffuse pancreatic tissues which present in the abdominal fat. Granulomas containing eggs and mononuclear and eosinophilic granular cell infiltrations, together with fat necrosis of the pancreatic tissue, were the most common pathological findings. Migrating larvae and peritoneal adhesions with organised fibrosis were also observed. In the infected tissues as well as in the spleen, liver and kidney, interleukin 1β (IL-1β) and cyclooxygenase (COX)-2 were upregulated than in control non-infected fish. Our results confirmed that H. japonica is infecting orange-spotted grouper caught from the Red Sea in Egypt with prominent tissue specificity in the mesentery, adipose tissue surrounding the intestine, and in the peritoneum.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The orange-spotted grouper, Epinephelus coioides, is an economically significant marine teleost (Chen et al. 2005). The colouration of a fish is produced by different colour pigments in epidermal and dermal layers of the skin which are largely contained within cells called chromatophores. The erythrophores have erythrin pigments (red), melanophores have melanin pigments (black) and xanthophores contain xanthin pigments (yellow) (Roberts 2012). However, when commercial grouper aquaculture expands, different stressors are increased as a result of high-density breeding, which creates negative changes in the environment and increases the susceptibility of fish to illnesses (Nagasawa and Cruz-Lacierda 2004). Wild and cultured grouper could be infected by different protozoan parasites, trematode and nematode which in turn makes the muscle of fish rejected by the consumer (Attia et al. 2021a, b).
Huffmanela spp. are a nematode that infects both marine and freshwater fishes; this genus is histozoic parasites that infect the skin, gills, muscle, swimbladder wall, serosa covering gut and bones, and cause a variety of clinical disease condition depending on the host and tissue (Esteves et al. 2016; Ramos et al. 2019; Eissa et al. 2021). This nematode was identified based on the shape of its eggs, which are trichuroid. The length and width of the eggs, as well as the egg surface, polar plug and egg envelop, are used to identify them. Huffmanela spp. could infect different marine fishes as recorded by Moravec (2001); Ramos et al. (2019) found that H. lusitana infects the muscles of pouting Trisopterus luscus, and H. paronai infects the skin of Xiphias gladius (Ramos et al. 2019). Moravec et al. (1998) found H. japonica and H. shikokuensis in the flesh of Upeneus bensasi and Stephanolepis cirrhifer in Japan. Moravec and Campbell (1991) found H. schouteni in the serosa protecting the colon, the abdominal cavity and the liver of Hirundichthys affinis and Cypselurus cyanopterus, respectively.
Varied Huffmanela spp. induce different alterations in fish, ranging from mild to severe darkening of the skin or muscle meat, which may be rejected by consumers (Ramos et al. 2019).
The zoonotic significance of Huffmanela spp.-infected fish muscles is unknown, although there have been numerous reports of Huffmanela spp. eggs in the stool of a Spanish child (Gállego et al. 1993) and from patients recorded by Schouten et al. (1968), who discovered H. schouteni eggs as a result of eating fish meals infected with Huffmanela spp.
The immune response to various stressors includes the release of various inflammatory mediators such as cytokines, which are small proteins that act as intercellular mediators to regulate the strength and duration of immunological responses. For example, active macrophages and blood monocytes release the pleiotropic cytokine interleukin (IL)-1β (Dinarello 1997; Bird et al. 2002; Apte et al. 2006), which are well-known for their critical role in inflammation and have the ability to activate a large number of non-structural, function-associated genes that are seldom expressed in healthy people. It also boosts the proliferation, growth and activity of non-specific (NK cells, macrophages and other immune cells) as well as specific immune cells (T and B cells; Apte et al. 2006; Dinarello 1996).
Therefore, this study aimed to identify and characterise the causative agent of the observed black spot lesions that noticed in the mesenteric tissues of orange-spotted grouper, Epinephelus coioides, and identification of the detected Huffmanela spp. parasite with studying its immunological and pathological effects on this examined fish, which caught from the Red Sea, Suez Governorate, Egypt.
Materials and methods
Fish samples
One hundred orange-spotted grouper were collected from different fish markets (freshly caught) at the Suez Governorate, which were caught from the Red sea; the fish weigh from 2 to 15 kg. All the collected fish were examined for any gross lesion in its muscle and/or its viscera. This study was approved from the Institutional Animal Care and Use Committee of Faculty of Veterinary Medicine with number: Vet Cu 8/03/2022/412.
Parasitological examination
Different portions of the peritoneal cavity with black strikes were examined in the Parasitology Department of Cairo University; Faculty of Veterinary Medicine. The peritoneum had a several black spots. The existence of the eggs, larvae or adult nematode was investigated using a stereoscopic microscope on wet-mounted slides without cover-slip pressure, and specimens from the muscles were studied using a light microscope.
The eggs in the peritoneum of the grouper were fixed in a formalin solution with a neutral buffered. The eggs were washed with lactophenol for an hour before being placed in glycerol jelly for light microscopy (Attia et al. 2021a, b). Photographs and measurements were collected from 20 eggs using an Olympus CX40 combined with a Nikon camera (Ruiz et al. 2013). The measurements were expressed as mean (minimum – maximum ± standard deviation) which was provided in micrometres (μm).
Scanning electron microscopy examination of Huffmanela spp. egg ultrastructure (SEM)
The infected grouper peritoneum was fixed for 24 h at 4 °C in 2.5% glutaraldehyde. The peritoneum was dehydrated by exposing it to a series of ethanol concentrations for 20 min each. The peritoneum was dehydrated using Whitman no.1 filter paper. A CO2 critical point drier was then used to finish the drying process (Autosamdri-815, Germany). Finally, the peritoneum samples were connected to the stubs and gold sputter coated at a thickness of 20 nm (Spi-Module sputter Coater, UK). All specimens were analysed and photographed at the Faculty of Agriculture, Cairo University, Egypt, using a JSM 5200 Electron Probe Microanalyzer from Joel, Japan. An electron microscope was used to picture the eggs, and a total of 20 eggs were examined (Ramos et al. 2019 and Attia et al. 2021a, b).
Egg morphology and measurements
Many types of eggs were discovered, stage 1 (light brown-shelled eggs with larvae) and stage 2 (dark brown-shelled eggs with larvae). Huffmanela spp. were identified based on the characteristics of advanced brown-shelled eggs with larvae (stages 1, 2), according to Justine (2005; 2007; 2011) and Justine and Iwaki (2014).
Gross lesions and histopathological examination
Observed gross lesions in the internal organs of infected fish were described, and a specimen from the peritoneum and different visceral organs was collected and fixed in 10% neutral buffered formalin for 24 h, then dehydrated in ascending grades of ethyl alcohol (50–100%), then cleared in xylene (2 change), then embedded in melted paraffin wax and stained by H&E (Roberts 2012).
Immunological cytokines analysis using real-time polymerase chain reaction (qRT-PCR)
Sampling
Aseptically dissected mesentery, peritoneum, liver, spleen and kidney of infected fish (0.5 cm2) were sampled from fish which had only the parasites under investigation. Same specimens from ten uninfected grouper samples were carefully examined for parasites, and these fish samples were gathered in the same way and served as negative controls.
The samples were homogenised with an MP Biomedicals FastPrep-24 Homogenizer (2 cycles of 30 s at 6 m/s). The data were then extracted using an RNA isolation kit (Ambion, Applied Biosystems). Five hundred nanograms of RNA was processed with DNase I amplification grade according to the manufacturer’s instructions (Invitrogen). Following the manufacturer’s procedure, the treated RNA was reverse-transcribed using the High-Capacity cDNA Archive Kit (Applied Biosystems) (Attia et al. 2021c; Botwright et al. 2021).
The PCR primer for orange-spotted grouper was created as recorded in the sequence deposited in the GenBank (Table 1); the reference gene utilised for sample normalisation was β-actin.
The RNA was extracted, synthesised and cycled using the cDNA synthesis procedures described by Attia et al. (2021c).
A Step OneTM Real-Time PCR System was used to perform quantitative PCR tests (Applied Biosystems, USA). One microliter of cDNA with 0.5 μl primer set (forward and reverse) was mixed in a reaction with SYBR® Premix Ex TaqTM, and the final volume was completed to 20 μl with ultra-pure water. The ∆CT value was obtained by subtracting the β-actin ∆CT as an internal control from the ∆CT value of the samples gene, where ∆CT is the cycle number; the PCR cycling conditions were followed as described by Botwright et al. (2021) and Attia et al (2021c). In each sample, the qRT-PCR was performed three times. The PCR cycling conditions were denaturation for 30 s at 94 °C, annealing for 30 s at 60 °C and extension for 45 s at 72 °C which were used in a 40-cycle amplification.
Statistical analysis
The one-way ANOVA test was used to compare different gene groups (SPSS Inc., Chicago, IL, USA). P < 0.05 was used to determine the significance of the data. Predictive Analytics Software (PASW) statistics, Version 18.0, was used for all statistical analysis (Attia et al. 2021d).
Results
Type-host—orange sponge grouper.
Site of infection—eggs deposited in the peritoneum.
Type-locality—Egypt.
Prevalence—5/100 (5%).
Etymology—Huffmanela japonica named as the eggs were first described in Japan.
Gross lesions of the infected fish
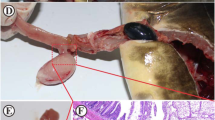
The external examination revealed no gross lesions (Fig. 1A) either on the skin or the gills. Internally, the lesions appeared as multifocal brown to black spots were noticed on the mesenteric fat and serosa surfaces of both the stomach and intestine. The black spots were also surrounding the diffuse pancreatic tissues in the mesentery. The pancreatic tissues showed swelling and light brown discoloration (Fig. 1B). Under dissecting microscope, the serosa surface of the visceral organs showed clusters of many characteristic Huffmanela spp. eggs (Fig. 1C and D); and the most of the infected fishes showed focal or diffuse peritoneal adhesions together with abundant fibrous connective tissue. Other organs including the liver, spleen, kidney and muscle were apparently normal and free of black spots.
A Orange-spotted grouper Epinephelus coioides without any external lesions. B Visceral organs and mesenteric tissues showing multifocal black spots on the mesenteric fat, serosa of stomach and intestine (arrows) and around the diffuse pancreatic tissues (p). C Serosa surface of the visceral organs of orange-spotted grouper under dissecting microscope is showing black spots with collection of eggs (arrows) in groups (× 4). D Higher magnification of the collected eggs (× 10)
Identification of Huffmanela spp. eggs using a light and scanning electron microscope
All of the eggs were demonstrated in the peritoneum and mesenteric tissues only, the eggs of the trichosomoidid genus Huffmanela. The results of light microscope (Figs. 2, 3, 4) and scanning electron microscope (Figs. 5 and 6) demonstrated the evidences of parasitological identification. The eggs are present in two stages (stage 1, light brown advanced eggs with larva; stage 2, dark brown advanced eggs with developed larva). Infection with H. japonica was found in all of the tested eggs. Stage 1 (fertilised eggs; n = 25): progressed eggs, light brown in colour, with a width of 32.5 (21–35 μm ± 0.5); the egg forms oval to spindle measuring 74.68 (65–78 μm ± 0.85) in length and with projecting polar plug.
The eggs had two layers: an outside brown layer and an inner clear layer; a transparent layer with eggshell thickness ranging from 3.8 (3.0–5.5 μm ± 0.97) and eggshell protuberance enclosed by a smooth egg envelop and a polar plug ranging from 7.9 (6.6–12 μm ± 1.8). The eggs contain convoluted larvae coiled within them, with a larval thickness of 5.9 (3.9–6.9 μm ± 0.9); stage 2 (fertilised eggs include larva; n = 25): more advanced eggs were brown to dark brown in colour; eggs were less broad with a width of 34.5 (25–35 μm ± 0.97). The egg shape was oval to spindle measuring 75.69 (65–78 μm ± 0.90) in length and with protruding polar plug. The eggs were made up of two layers: an outside brown layer and an interior transparent layer with eggshell thickness ranged from 4.3 (4.0–5.5 μm ± 0.87) and with eggshell protuberance which surrounded with smooth egg envelop; polar plug ranged from 7.9 (6.8–12 μm ± 2.8); the eggs contained convoluted larvae coiled within them, with a larval thickness of 6.0 (4.9–7.9 μm ± 0.9) (Table 2).
Histopathological findings
The histopathological examination of infected fish showed focal aggregation of the characteristic shape, H. japonica eggs with brown, thick egg shell. The lesions were noticed in the mesenteric fat, serosa surface of the stomach and intestine, and surrounding the pancreatic tissues. The tissues of other internal organs were free of H. japonica eggs, but with mild activation of melanomacrophage centres in the spleen. The H. japonica eggs were embedded in the tissue. One egg (Fig. 7A), binaries or multiple eggs were aggregated in the mesenteric tissue forming granulomatous reaction (Fig. 7B, C and D). The granulomas consist of eggs surrounded with fibrous connective tissue capsule, oedema and mononuclear inflammatory cells such as lymphocytes and macrophages. The eggs within the granuloma were contained prominent larvae, but sometimes, they were degenerated and appeared homogenous and eosinophilic without internal structures. Pancreatic fat necrosis was also detected (Fig. 8A); in such cases, eosinophilic granular cell infiltration was noticed around the granulomas (Fig. 8B and C). In a few cases, cross sections of nematode larvae were observed in the serosa of the intestinal tissues (Fig. 8D).
Photomicrograph for histopathological sections of infected Epinephelus coioides with H. japonica showing A degenerated egg of H. japonica (eg) deeply embedded in mesenteric fat (mf) near the muscular layer (m) of the intestine; the pancreatic tissue (p) around the hepatic vein branch (v) showed fat necrosis. B Serosa surface of the intestine showing multifocal granulomatous reactions. C Granuloma (g) containing multiple H. japonica eggs surrounded with dissociated fibrous tissue with oedema. D Granuloma containing multiple H. japonica eggs; some eggs were degenerated (d) without prominent internal structures and other eggs (eg) have larvae. The granuloma surrounded with abundant fibrous connective tissue capsule (f)
Photomicrograph for histopathological sections of infected Epinephelus coioides with H. japonica showing A multiple granulomatous reactions (g) in the mesenteric tissue. B Advanced lesion showing cross and sagittal sections of many H. japonica eggs (eg) within the granulomas (g) and pronounced fibrosis (f). C Marked mononuclear inflammatory cells (ic) and eosinophilic granular cell infiltration (egc) surrounding the granulomas (g). D Cross section in the migrating H. japonica larva in the abdominal cavity near the muscular layer (m) of the intestine
Expression analysis of the interleukin-1β (IL-1β) and cyclooxygenase (COX)-2 through using of quantitative real-time PCR from different infected organs
In comparison of the serosa mesenteric tissue, peritoneum and other internal organs of infected and non-infected orange-spotted grouper by Huffmanela japonica, the IL-1ß was 24 folds upregulated in peritoneum of infected fish than in control noninfected one, while its expression of the spleen was 19 folds upregulated than in control non-infected fish. The liver and kidney were lower in upregulation than in other infected organs (Fig. 9). All genes were expressed significantly higher than the control group (P = 0.0001).
Dealing with cyclooxygenase (COX)-2 expressions (Fig. 10), in the infected tissues of orange-spotted grouper by Huffmanela japonica, (COX)-2 was 30 folds upregulated than in control non-infected fish. Concerning the expression of the spleen, (COX)-2 was 25 folds upregulated than in control non-infected fish, while the liver and kidney were lower in upregulation than in other infected organs. All genes were expressed significantly higher than the control group (P = 0.0001).
Discussion
Huffmanela spp. eggs were histozoic which includes intracellular nematodes (Moravec 1987; Moravec et al. 1998) that are typically classified based on the morphology of their eggs and larvae (Table 3); with only 5 of the 17 accepted species, 29% (Carballo and Navone 2007) have descriptions that include information on the adult male or female; gill and skin epithelia, skeletal muscle, serosa and mesentery, swim bladder and bone are all infected by adults and eggs (Moravec and Garibaldi 2000).
Gills and skin epithelia, skeletal muscle, serosa and mesentery, swim bladder and bone are all infected by adults and eggs (Moravec 1987, 2001). The number of Huffmanela species described in recent years has risen, indicating that these parasites are likely to be widespread, particularly among marine fishes. Unidentified Huffmanela eggs were recently found in the stool of a youngster in Spain (Gállego et al. 1993), presumably as a result of consuming infected fish previously. H. schouteni eggs were found in the stools of six patients in a similar case.
Although there are few characteristics that may be used to distinguish eggs, many diagnostic features have been created and used to separate these species based on the egg morphology (Moravec and Fajer-Avila’s 2000).
Based on previously published data, there are approximately 21 species of Huffmanela that have been recorded around the world to date; these species were identified according to Moravec and Fajer-Avila’s (2000) with their width; shape of shell smooth or spiny with ridges or filaments; with envelop or not; the envelop character which is spinose or aspinose; and the habitat of eggs which is the skin, gills, muscles, swimbladder and within bones.
As a result of the research conducted by Attia et al. (2021a) on an orange-spotted grouper (Epinephelus coioides), the fish was rejected in the fish market due to extensive black spots on its viscera which revealed presence of Huffmanela; the internal infection of orange-spotted grouper with H. japonica which observed in this study may affect fish marketability. The length and breadth of the eggs were substantially identical to those found in a previous research by Moravec et al. (1998), who measured H. japonica egg length at 58–69 × 26–30 μm. According to Moravec et al. (1998), the egg shell in our investigation displayed protuberance; in addition, the eggshell envelop was smooth, as demonstrated by scanning electron microscopy; Moravec et al. (1998) validated this observation.
Other results in H. huffmani had spines on eggshell; these spines seem triangular as recorded in ultrastructure of transmission electron microscope by Žd’árská et al. (2001), but these spines did not resemble those in our study. The protuberance did not appear in this research in the same way as it did in H. huffmani, which had a consistent form and distribution. H. hamo was found in the swimbladder of freshwater fishes (Huffman and Moravec 1988), whereas our research was found in the peritoneum of marine fishes of orange-spotted grouper.
While Moravec et al. (1998) identified the eggs present in the musculature of the infected goatfish Upeneus bensasi (Mullidae: Perciformes), the eggs clustered present in groups of the affected muscles. In our study, this is the first to characterise these eggs using a scanning electron microscope as Attia et al. (2021a), which was not observed in Moravec et al. (1998) results, while in Attia et al. (2021a), they found the H. japonica in the muscles of orange-spotted grouper in Saudi Arabia. In contrast to this study, we observed the black discolouration in peritoneum not in muscles.
While the presence of H. schouteni in the intestinal serosa of fish by Moravec and Campbell (1991) was discovered in flying fish in Curaçao, but the eggs had envelop with protuberance not smooth and the eggs had a smooth shell in contrast to our study that the eggs had smooth envelop with protuberance egg shell.
In our study, the main pathological findings of the infected fish showed tissue reaction against Huffmanela eggs. The lesions were observed in the mesentery, adipose tissue and its associated diffuse pancreatic tissues; grouper spp. have a scattered diffuse pancreas in the mesenteries and viscera within the peritoneal cavity (Adel 2013). The observed granulomatous reaction, mononuclear inflammatory cells and eosinophilic granular cell infiltration, together with necrosis of the pancreatic tissue, were the most common findings in our study, and agreed with Attia et al. (2021a) who recorded granulomatous reaction in the skeletal muscles of orange-spotted grouper (Epinephelus coioides) infected with Huffmanela spp. Although the records of Huffmanela infection in Epinephelus fishes are scarce, lots of studies recorded Huffmanela egg infection in different other fishes, with prominent host and tissue specificity (Huffman and Moravec 1988; Cox et al. 2004; Moravec et al. 2005; Carballo and Navone 2007; Bullard et al. 2012; Justine and Iwaki 2014; Esteves et al. 2016; Ramos et al. 2019; Eissa et al. 2021; Attia et al. 2021b).
Regarding the immune reaction of the H. japonica in this study; the two expressed genes were elevated in different organs, especially the infected serosa and peritoneum while in uninfected organs, spleen, kidney and liver; due to the presence of the most immunological genes in the spleen; with lowering in kidney and liver.
Furthermore, with the availability of fish recombinant IL-1, the biological activities of fish IL-1 were partially characterised in vitro and in vivo (Hong et al. 2001; Buonocore et al. 2004; 2005). These studies revealed that, like their mammalian counterparts, fish IL-1 affected cell proliferation and phagocytosis, as well as promoted the expression of immune-related genes such as cyclooxygenase (COX)-2 and MHC class II chain (Buonocore et al. 2005; Hong et al. 2001; 2004).
So, our findings may add new information for the existence of Huffmanela infection in orange-spotted grouper, specifically in the mesenteric and pancreatic tissue. In this concern, the host-parasite interaction is a multifactorial condition, and the existence of the parasite could be attributed to age, environmental change, host behaviour and expression of immune-regulating genes (Khan. 2012 and Botwright et al. 2021). The observed inflammatory cells including macrophages, lymphocytes and eosinophilic granular cells are the tissue reaction against Huffmanela parasitism in many fishes and considered immune response against this type of nematodes. Both humoral and cellular immune reactions are prominent in fish endoparasitic nematode infection (Buchmann 2012).
Conclusion
H. japonica is infecting orange-spotted grouper which is caught from the Red Sea, Egypt, with prominent black spots, discoloration and tissue specificity in the mesenteric tissues and peritoneum. It is recommended to find the possible way to treat this fish in the future research which needs more efforts economically and in the field.
Data availability
All the authors declare that all the data supporting the results reported in our article were found and included in this article only.
Change history
05 July 2023
A Correction to this paper has been published: https://doi.org/10.1007/s10499-023-01182-9
Abbreviations
- IL-1β:
-
Interleukin 1β
- NK:
-
Natural killer cell
- qRT-PCR:
-
Quantitative real-time polymerase chain reaction
- (COX)-2:
-
Cyclooxygenase-2
- MHC:
-
Major histocompatibility complex
References
Adel AH (2013) Anatomy and Histology of the digestive system of the carnivorous fish, the brown-spotted Grouper, Epinephelus chlorostigma (Pisces; Serranidae) from the Red Sea. Life Sci J 10(2):2149–2164
Apte RN, Dotan S, Elkabets M, White MR, Reich E, Carmi Y, Song X, Dvozkin T, Krelin Y, Voronov E (2006) The involvement of IL-1 in tumorigenesis, tumor invasiveness, metastasis and tumor–host interactions. Cancer Metastasis Rev 25:387–408
Attia MM, Mahmoud MA, Ibrahim MM (2021a) Morphological and pathological appraisal of Huffmanela sp. (Nematoda: Trichosomoididae) infecting orange-spotted grouper (Epinephelus coioides, Hamilton, 1822) at Jubail Province, Saudi Arabia: a case report. J Parasit Dis.: Official organ of the Indian Society for Parasitology 45(4):980–985
Attia MM, Ibrahim MM, Mahmoud MA, Al-Sabi MNS (2021b) Huffmanela sp. (Nematoda: Trichosomoididae: Huffmanelinae) Encountered in the Whitecheek Shark (Carcharhinus Dussumieri) in The Arabian Gulf. Helminthologia 58(3):281–291.
Attia MM, Elgendy MY, Abdelsalam M et al (2021c) Morpho-molecular identification of Heterophyes heterophyes encysted metacercariae and its immunological and histopathological effects on farmed Mugil cephalus in Egypt. Aquacult Int 29:1393–1407
Attia MM, Abdelsalam M, Korany RM, Mahdy OA (2021d) Characterization of digenetic trematodes infecting African catfish (Clarias gariepinus) based on integrated morphological, molecular, histopathological, and immunological examination. Parasitol Res 120(9):3149–3162
Bird S, Zou J, Wang T, Munday B, Cunningham C, Secombes CJ (2002) Evolution of interleukin-1β. Cytokine Growth Factor Rev 13:483–502
Botwright NA, Mohamed AR, Slinger J, Lima PC, Wynne JW (2021) Host-Parasite Interaction of Atlantic salmon (Salmo salar) and the Ectoparasite Neoparamoeba perurans in Amoebic Gill Disease. Front Immunol 31(12):672700
Buchmann K (2012) Fish immune responses against endoparasitic nematodes – experimental models, review article. J Fish Dis 35:623–635
Bullard SA, Ruiz CF, McElwain A, Murray MJ, Borucinska JD, Benz GW (2012) Huffmanela cf. carcharhini (Nematoda: Trichosomoididae: Huffmanelinae) from skin of a sandbar shark, Carcharhinus plumbeus, in the Pacific Ocean. J Parasitol 98:333–340
Buonocore F, Mazzini M, Forlenza M, Randelli E, Secombes CJ, Zou J, Scapigliati G (2004) Expression in Escherichia coli and purification of sea bass (Dicentrarchus labrax) interleukin-1β, a possible immuno-adjuvant in aquaculture. Mar Biotechnol (NY) 6:53–59
Buonocore F, Forlenza M, Randelli E, Benedetti S, Bossu P, Meloni S, Secombes CJ, Mazzini M, Scapigliati G (2005) Biological activity of sea bass (Dicentrarchus labrax L.) recombinant interleukin-1β. Mar Biotechnol (NY) 7:609–617
Carballo MC, Navone GT (2007) A new Huffmanela species (Nematoda: Trichosomoididae) parasitizing atherinid fishes in North Patagonian Gulfs. Argentina J Parasitol 93:377–382
Chen R, Li W, Lin H (2005) cDNA cloning and mRNA expression of neuropeptide Y in orange spotted grouper Epinephelus coioides. Comp Biochem Physiol b: Biochem Mol Biol 142:79–89
Cox MK, Huffman DG, Moravec F (2004) Observations on the distribution and biology of Huffmanela huffmani (Nematoda: Trichosomoididae). Folia Parasitol 51:50
Dinarello CA (1996) Biologic basis for interleukin-1 in disease. Blood 87:2095–2147
Dinarello CA (1997) Interleukin-1. Cytokine Growth Factor Rev 8:253–265
Eissa IAM, Gadallah AO, Hashim M, Noureldin EA, Bayoumy EM, Haridy M (2021) First record of the nematode, Huffmanela sp. infecting the broomtail wrasse (Cheilinus lunulatus) from Egypt. J Parasit Dis 45:228–235
Esteves A, Oliveira I, Ramos P, Carvalho A, Nazario N, Seixas F (2016) Huffmanela spp. (Nematoda, Trichosomoididae) from Microchirus azevia: tissue location and correspondence of host muscle discoloration with parasite burden. J Fish Aquat Sci 11:304–310
Gállego J, Riera C, Portús M (1993) Huffmanela sp. eggs (Nematoda: Trichosomoididae), as a human spurious parasite in a child from Barcelona (Spain). Folia Parasitol 40:208–210
Hong S, Zou J, Crampe M, Peddie S, Scapigliati G, Bols N, Cunningham C, Secombes CJ (2001) The production and bioactivity of rainbow trout (Oncorhynchus mykiss) recombinant IL-1 beta. Vet Immunol Immunopathol 81:1–14
Hong S, Zou J, Collet B, Bols NC, Secombes CJ (2004) Analysis and characterization of IL-1 beta processing in rainbow trout Oncorhynchus mykiss. Fish Shellfish Immunol 16:453–459
Huffman DG, Moravec F (1988) First description of adult Huffmanela huffmani Moravec, 1987 (nematode: Trichosomoididae) from the swimbladder of centrarchid fishes of the upper San Marcos River, Central Texas. Folia Parasitol 35:227–234
Justine JL (2004) Three new species of Huffmanela Moravec, 1987 (Nematoda: Trichosomoididae) from the gills of marine fish off New Caledonia. Syst Parasitol 59:29–37
Justine JL (2005). Huffmanela lata n. sp. (Nematoda: Trichosomoididae: Huffmanelinae) from the shark Carcharhinus amblyrhynchos (Elasmobranchii: Carcharhinidae) off New Caledonia. Syst Parasitol 61:181–184
Justine JL (2007) Huffmanela spp. (Nematoda, Trichosomoididae) parasites in coral reef fishes off New Caledonia, with descriptions of H. balista n. sp. and H. longa n. sp. Zootaxa 1628:23–41
Justine JL (2011) Huffmanela plectropomi n. sp. (Nematoda: Trichosomoididae: Huffmanelinae) from the coral grouper Plectropomus leopardus (Lacépède) off New Caledonia. Syst Parasitol 79:139–143
Justine JL, Iwaki T (2014) Huffmanela hamo sp. n. (Nematoda: Trichosomoididae: Huffmanelinae) from the dagger-tooth pike conger Muraenesox cinereus off Japan. Folia Parasitol 61:267–271
Khan RA (2012) Host-parasite interactions in some fish species; review article. J Parasitol Res 237280:7
Moravec F (1987) Revision of Capillarità nematodes (subfamily Capillariinae) parasitic in fishes. Studie ČSAV No. 3. Academia, Praha, Českě Budějovice, Czech Republic, p 141
Moravec F (2001) Trichinelloid nematodes parasitic in cold blooded vertebrates. Academia, Praha, p 429
Moravec F, Campbell BG (1991) A new Huffmanela species, H. schouteni sp. n. (Nematoda, Trichosomoididae), from flying fishes in Curaçao. Folia Parasitol 38:29–32
Moravec F, Fajer-avila E (2000) A new Huffmanela species, H. mexicana n. sp. (Nematoda: Trichosomoididae), from the marine fish Sphoeroides annulatus in Mexico. J Parasitol 86:1229–1231
Moravec F, Garibaldi F (2000) Huffmanela paronai sp. n. (Nematoda: Trichosomoididae), a new parasite from the skin of swordfish Xiphias gladius in the Ligurian Sea (Western Mediterranean). Folia Parasitol 47:309–313
Moravec F, Koudela B, Ogawa K, Nagasawa K (1998) Two new Huffmanela species, H. japonica n. sp. and H. shikokuensis n. sp (Nematoda: Trichosomoididae), from marine fishes in Japan. J Parasitol 84:589–593
Moravec FE, Conboy GA, Speare J (2005) A new trichosomoidid from the skin of Sebastes spp. (Pisces) from British Columbia, Canada. J Parasitol 91:411–414
Nagasawa K, Cruz-Lacierda ER (2004) Diseases of cultured groupers. Southeast Asian Fisheries Development Center Aquaculture Department, pp 40–65
Ramos P, Carvalho R, Rosa F, Alexandre-Pires G, Seixas F, Esteves A, and Huffman D (2019) Huffmanela lusitana sp. n. (Nematoda: Trichosomoididae) infecting pouting, Trisopterusluscus (Teleostei: Gadidae) off the Atlantic coast of Portugal. Int J Parasitol Parasites Wildl 9:266–273
Roberts RJ (2012) Fish Pathology, 3rd edn. W.B. Saunders, Philadelphia, PA
Ruiz CF, Bullard SA (2013) Huffmanela markgracei sp. n. (Nematoda: Trichosomoididae) from buccal cavity of Atlantic sharpnose shark, Rhizoprionodon terraenovae (Carcharhiniformes: carcharhinidae), in the northwestern Gulf of México off Texas. Folia Parasitol 60:353–358
Ruiz CF, Ray CL, Cook M, Grace MA, Bullard SA (2013) A new species of Trichosomoididae (Nematoda) from skin of red snapper, Lutjanus campechanus (Perciformes: Lutjanidae), on the Texas-Louisiana shelf, northern Gulf of Mexico. J Parasitol 99:318–326
Schouten H, Suriel-Smeets RM, Kibbelaar MA (1968) The simultaneous occurrence of ova resembling Dicrocoelium dendriticum or Capillaria hepatica in the stools of inhabitants of Curaçao. Trop Geogr Med 20:271–275
Žd’árská Z, Huffman DG, Moravec F, Nebesářová J (2001) Egg shell ultrastructure of the fish nematode Huffmanela huffmani (Trichosomoididae). Folia Parasitol 48:231–234
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study’s conception and design. MA (Marwa M. Attia) conceived and designed the study. MA and MI performed the research, data collection and analysis. Material preparation and molecular design were performed by MA; pathological analysis was performed by MA (Mahmoud Aly) and MI. All authors write the manuscript and revised it. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval and consent to participate
This study was approved and follows the guidelines of the ethical committee of Cairo University, Faculty of Veterinary Medicine; these experiments were performed in compliance with the ARRIVE guidelines.
Consent for publication
All data have no human data or results.
Conflict of interest
The authors declare no competing interests.
Additional information
Handling Editor: Amany Abbass
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Attia, M.M., Ibrahim, M.M. & Mahmoud, M.A. Heavy infection of the orange-spotted grouper (Epinephelus coioides) with Huffmanela japonica: morphological, ultrastructural identification, tissue reactions and immunological analysis. Aquacult Int 31, 3311–3327 (2023). https://doi.org/10.1007/s10499-023-01124-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-023-01124-5