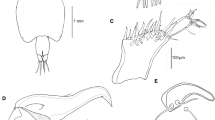
Abstract
Members of the family Lamippidae are obligate endoparasitic copepods that typically associate with octocorals. In the present work, the association between Lamippe bouligandi and its bathyal sea pen host Anthoptilum grandiflorum was examined. A total of 1463 females and 23 males of L. bouligandi were collected from 195 colonies of A. grandiflorum. Details of the external morphology and digestive organs of the female copepod are used to refine our understanding of its feeding strategy and relationship with the host, and a preliminary overview of the male is provided for the first time. A combination of histology, scanning electron microscopy and laboratory monitoring of live sea pens hosting L. bouligandi were used to elucidate aspects of its life history, including the reproductive cycle, brooding strategy, and embryonic and larval development.









Similar content being viewed by others
Data availability
Gene sequences in GenBank, specimens deposited in museum.
Code availability
Not applicable.
References
Anstensrud M (1990) Mating strategies of two parasitic copepods [(Lernaeocera branchialis (L.) (Pennellidae) and Lepeophtheirus pectoralis (Müller) (Caligidae)] on flounder: polygamy, sex-specific age at maturity and sex ratio. J Exp Mar Biol Ecol 136:141–158. https://doi.org/10.1016/0022-0981(90)90192-F
Appeltans W, Ahyong ST, Anderson G et al (2012) The magnitude of global marine species diversity. Curr Biol 22:2189–2202. https://doi.org/10.1016/j.cub.2012.09.036
Arnaud J, Brunet M, Mazza J (1980) Structure et ultrastructure comparées de l’intestin chez plusieurs espèces de copépodes Calanoides (Crustacea). Zoomorphologie 95:213–233
Baillon S, Hamel J-F, Wareham VE, Mercier A (2012) Deep cold-water corals as nurseries for fish larvae. Front Ecol Environ 10:351–356. https://doi.org/10.1890/120022
Baillon S, Hamel J-F, Mercier A (2014a) Diversity, distribution and nature of faunal associations with deep-sea pennatulacean corals in the Northwest Atlantic. PLoS ONE 9:e111519. https://doi.org/10.1371/journal.pone.0111519
Baillon S, Hamel J-F, Wareham Hayes V, Mercier A (2014b) Seasonality in reproduction of the deep-water pennatulacean coral Anthoptilum grandiflorum. Mar Biol. https://doi.org/10.1007/s00227-013-2311-8
Bouligand Y (1960a) Sur l’oganisation des Lamippides, copépodes parasites des octocoralliaires (I). Première. Note Vie Milieu 11:355–380
Bouligand Y (1960b) Notes sur la famille des Lamippidae, Première Partie. Crustaceana 1:258–278
Bouligand Y (1961) Notes sur la famille des Lamippidae, 2e Partie. Crustaceana 2:50–52
Boxshall GA (2005) Crustacean parasites (Copepoda). In: Rohde K (ed) Marine parasitology. CABI, Oxon, p 123–138
Briggs RP (1977) Structural observations on the alimentary canal of Paranthessius anemoniae, a copepod associate of the snakelocks anemone Ammonia sulcata. J Zool 182:353–368. https://doi.org/10.1111/j.1469-7998.1977.tb03915.x
Bruzelius R (1859) Über einen in der Pennatula rubra lebenden Schmarotzer (Lamippe). (Mit 1 Taf.). Arch F Naturg 25:286–290
Buhl-Mortensen L, Mortensen PB (2004a) Symbiosis in deep-water corals. Symbiosis 37:33–61
Buhl-Mortensen L, Mortensen PB (2004b) Gorgonophilus canadensis n. gen., n. sp. (Copepoda: Lamippidae), a gall forming endoparasite in the octocoral Paragorgia arborea (L., 1758) from the Northwest Atlantic. Symbiosis 37:155–168
Castro P (1988) Animal symbioses in coral reef communities: a review. Symbiosis 5:161–184
Claparède JV (1867) Lamippe proteus. Ann Sci Nat Ser 5(8):23–28
Cordeiro R, McFadden C, van Ofwegen L, Williams G (2020) World List of Octocorallia. Anthoptilum grandiflorum (Verrill, 1879). Accessed through: World Register of Marine Species at: http://www.marinespecies.org/aphia.php?p=taxdetails&id=128504#distributions. Accessed 9 Nov 2020
Cornwall IE (1927) Some North Pacific whale barnacles. Contri Can Biol 3:501–517
Daase M, Søreide JE, Martynova D (2011) Effects of food quality on naupliar development in Calanus glacialis at subzero temperatures. Mar Ecol Prog Ser 429:111–124. https://doi.org/10.3354/meps09075
deWaard JR, Ivanova NV, Hajibabaei M, Hebert PDN (2008) Assembling DNA barcodes. In: Martin CC, Martin CC (eds) Environmental genomics. Humana Press, Totowa, pp 275–294
Dudley PL (1973) Enalcyonium carrikeri, a new species of lamippid copepod from Alcyonium carneum Agassiz in New England. Crustaceana 25:75–87
Grygier MJ (1983) An endoparasitic lamippid copepod in Acanella from the North Atlantic. Crustaceana 45:176–182
Hamel J-F, Mercier A (1994) New distribution and host record for the starfish parasite Dendrogaster (Crustacea: Ascothoracida). J Mar Biol Assoc UK 74:419–425. https://doi.org/10.1017/S0025315400039436
Hartog MM (1888) The morphology of cyclops and the relations of the Copepoda. Trans Linn Soc Lond 2nd Ser Zool 5:1–46. https://doi.org/10.1111/j.1096-3642.1888.tb00157.x
Ho J-S (1984) New family of poecilostomatoid copepods (Spiophanicolidae) parasitic on polychaetes from Southern California, with a phylogenetic analysis of Nereicoliform families. J Crustac Biol 4:134–146. https://doi.org/10.2307/1547902
Ho J-S, Jayarajan P, Radhakrishnan S (1992) Copepods of the family Ergasilidae (Poecilostomatoida) parasitic on coastal fishes of Kerala, India. J Nat Hist 26:1227–1241
Humes AG (1986) Myicola metisiensis (Copepoda: Poecilostomatoida), a parasite of the bivalve Mya arenaria in eastern Canada, redefinition of the Myicolidae, and diagnosis of the Anthessiidae n. fam. Can J Zool 64:1021–1033. https://doi.org/10.1139/z86-152
Humes AG (1994) How many copepods? Hydrobiologia 292:1–7. https://doi.org/10.1007/BF00229916
Humphrey CD, Pittman FE (1974) A simple methylene blue-azure II-basic fuchsin stain for epoxy-embedded tissue sections. Stain Technol 49:9–14. https://doi.org/10.3109/10520297409116929
Ivanova NV, Dewaard JR, Hebert PDN (2006) An inexpensive, automation-friendly protocol for recovering high-quality DNA. Mol Ecol Notes 6:998–1002. https://doi.org/10.1111/j.1471-8286.2006.01428.x
Jungersen HPE (1904) Pennatulida. Dan Ingolf-Exped 5:1–91
Kabata Z (1982) Copepoda (Crustacea) parasitic on fishes: problems and perspectives. In: Lumsden WHR, Muller R, Baker JR (eds) Advances in parasitology. Academic Press, Cambridge, pp 1–71
Kabata Z (1992) Copepoda parasitic on Australian fishes, XV. Family Ergasilidae (Poecilostomatoida). J Nat Hist 26:47–66
Korzhavina OA, Reimer JD, Ehrlich H, Ivanenko VN (2021) Global diversity and distribution of Lamippidae copepods symbiotic on Octocorallia. Symbiosis. https://doi.org/10.1007/s13199-021-00750-y
Laubier L (1972) Lamippe (Lamippe) bouligandi sp. nov., Copépode parasite d’Octocoralliaire de la Mer du Labrador. Crustaceana 22:285–293
Madanire-Moyo GN, Avenant-Oldewage A (2013) On the development of a parasitic copepod, Lamproglena clariae Fryer, 1956 (Copepoda, Lernaeidae) infecting the sharp tooth catfish, Clarias gariepinus. Crustaceana 86:416–436. https://doi.org/10.1163/15685403-00003165
Margolis L (1954) List of the parasites recorded from sea mammals caught off the west coast of North America. J Fish Res Board Can 11:267–283. https://doi.org/10.1139/f54-017
Nylund A, Økland S, Bjørknes B (1992) Anatomy and ultrastructure of the alimentary canal in Lepeophtheirus salmonis (Copepoda: Siphonostomatoida). J Crustac Biol 12:423–437. https://doi.org/10.2307/1549036
Patton WK (1972) Studies on the animal symbionts of the gorgonian coral, Leptogorgia virgulata (Lamarck). Bull Mar Sci 22:419–431
Ratnasingham S, Hebert PDN (2007) Bold: the barcode of life data system (http://www.barcodinglife.org). Mol Ecol Notes 7:355–364. https://doi.org/10.1111/j.1471-8286.2007.01678.x
Russell FS (ed) (1967) Chapter 9 Parasites of commercially important marine molluscs: the class Crustacea. In: Advances in marine biology. Academic Press, Cambridge, pp 286–335
Sabatini DD, Bensch K, Barrnett RJ (1963) Cytochemistry and electron microscopy: the preservation of cellular ultrastructure and enzymatic activity by aldehyde fixation. J Cell Biol 17:19–58. https://doi.org/10.1083/jcb.17.1.19
Stein M (2007) Oceanography of the Flemish cap and adjacent waters. J Northwest Atl Fish Sci 37:135–146
Vader W (1970) Antheacheres duebeni M. Sars, a copepod parasitic in the sea anemone, Bolocera tuediae (Johnston). Sarsia 43:99–106. https://doi.org/10.1080/00364827.1970.10411172
Vehmaa A, Brutemark A, Engström-Öst J (2012) Maternal effects may act as an adaptation mechanism for copepods facing pH and temperature changes. PLoS ONE. https://doi.org/10.1371/journal.pone.0048538
Williams JD, Anchaluisa B, Boyko CB, McDaniel N (2018) Description of a new endoparasitic copepod genus and species (Lamippidae) that induces gall formation in leaves of the sea pen Ptilosarcus gurneyi (Octocorallia) from British Columbia. Mar Biodivers 48:1325–1335. https://doi.org/10.1007/s12526-016-0593-z
Wilson CH, Hartline DK (2011) Novel organization and development of copepod myelin. i. ontogeny. J Comp Neurol. https://doi.org/10.1002/cne.22695
Zulueta A (1908) Note préliminaire sur la famille des Lamippides, copépodes parasites des alcyonaires. Arch Zool Exp Gén 4:1–30
Zulueta A (1910) Deuxième note sur la famille des Lamippides. Arch Zool 6:147–148
Acknowledgements
We would like to thank the scientific staff of the In the methods we use Fisheries and Oceans Canada (DFO) and the Coast Guard crew aboard the CCGS Teleost for assistance during collection of the sea pens and copepods, and Igor Eeckhaut (Mons University, Belgium) for helping with the SEM and some histological analyses. We would also like to thank two anonymous reviewers for comments on earlier versions of the manuscript.
Funding
Natural Sciences and Engineering Research Council of Canada (NSERC), Canada Foundation for Innovation (CFI) and Mitacs from AM.
Author information
Authors and Affiliations
Contributions
Project development: AM and JFH; Material preparation: SB and JP; SEM and histology: JP; Investigation: HDP, SB, JP and JFH; Writing—original draft preparation: HDP, JFH and AM; Writing—reviewing and editing: all authors. Funding: AM.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
The authors consent to publication.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Penney, H.D., Baillon, S., Hamel, JF. et al. Morphology and biology of the endoparasitic copepod Lamippe bouligandi from the bathyal sea pen Anthoptilum grandiflorum. Symbiosis 85, 233–248 (2021). https://doi.org/10.1007/s13199-021-00811-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13199-021-00811-2