Abstract
It is well known that oocyte donation (OD) pregnancies are associated with higher complication rates compared to autologous pregnancies. However, evidence-based information for pre-pregnancy counseling designed for health care workers is scarce. Therefore, a systematic literature search was performed to find articles that address pre-pregnancy counseling before OD.
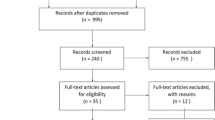
A systematic search was conducted in September 2020 in various databases, including PubMed and Embase. Nine (systematic) reviews and meta-analyses were included that reported on pre-pregnancy advice in OD pregnancies.
Studies are consistent in documenting a higher risk for hypertensive disorders, cesarean section, preterm birth, postpartum hemorrhage, and low birth weight. Based on these complications, pre-pregnancy advice is mentioned in all included systematic reviews to prevent complications in the next pregnancy. All studies recommend counseling women on the increased risk of complications during OD pregnancy. Other recommendations include the prophylactic use of aspirin in pregnancy and restriction to single embryo transfer. Individualized appropriate surveillance and management strategies should be considered for every patient achieving pregnancy by OD.
In conclusion, we provide a summary of the most important outcomes in OD pregnancies, and thereby offer a guide for pre-pregnancy counseling.


Similar content being viewed by others
Availability of Data and Material
Not applicable.
Code Availability
Not applicable.
References
Shufaro Y, Schenker JG. The risks and outcome of pregnancy in an advanced maternal age in oocyte donation cycles. J Matern Fetal Neonatal Med. 2014;27(16):1703–9. https://doi.org/10.3109/14767058.2013.871702.
Saito S, Nakabayashi Y, Nakashima A, Shima T, Yoshino O. A new era in reproductive medicine: consequences of third-party oocyte donation for maternal and fetal health. Seminars in immunopathology. 2016;38(6):687–97. https://doi.org/10.1007/s00281-016-0577-x.
Savasi VM, Mandia L, Laoreti A, Cetin I. Maternal and fetal outcomes in oocyte donation pregnancies. Hum Reprod Update. 2016;22(5):620–33. https://doi.org/10.1093/humupd/dmw012.
Kushnir VA, Gleicher N. Fresh versus cryopreserved oocyte donation. Curr Opin Endocrinol Diabetes Obes. 2016;23(6):451–7. https://doi.org/10.1097/med.0000000000000290.
Sagi-Dain L, Sagi S, Dirnfeld M. The effect of paternal age on oocyte donation outcomes. Obstet Gynecol Surv. 2016;71(5):301–6. https://doi.org/10.1097/ogx.0000000000000311.
Kamath MS, Sunkara SK. Perinatal outcomes after oocyte donation and in-vitro fertilization. Curr Opin Obstet Gynecol. 2017;29(3):126–30. https://doi.org/10.1097/gco.0000000000000356.
van der Hoorn ML, Lashley EE, Bianchi DW, Claas FH, Schonkeren CM, Scherjon SA. Clinical and immunologic aspects of egg donation pregnancies: a systematic review. Hum Reprod Update. 2010;16(6):704–12. https://doi.org/10.1093/humupd/dmq017.
Sagi-Dain L, Sagi S, Dirnfeld M. Effect of paternal age on reproductive outcomes in oocyte donation model: a systematic review. Fertil Steril. 2015;104(4):857-65.e1. https://doi.org/10.1016/j.fertnstert.2015.06.036.
Blazquez A, García D, Vassena R, Figueras F, Rodriguez A. Risk of pre-eclampsia after fresh or frozen embryo transfer in patients undergoing oocyte donation. Eur J Obstet Gynecol Reprod Biol. 2018;227:27–31. https://doi.org/10.1016/j.ejogrb.2018.05.030.
Pecks U, Maass N, Neulen J. Oocyte donation: a risk factor for pregnancy-induced hypertension: a meta-analysis and case series. Dtsch Arztebl Int. 2011;108(3):23–31. https://doi.org/10.3238/arztebl.2011.0023.
Masoudian P, Nasr A, de Nanassy J, Fung-Kee-Fung K, Bainbridge SA, El Demellawy D. Oocyte donation pregnancies and the risk of preeclampsia or gestational hypertension: a systematic review and metaanalysis. Am J Obstet Gynecol. 2016;214(3):328–39. https://doi.org/10.1016/j.ajog.2015.11.020.
Jeve YB, Potdar N, Opoku A, Khare M. Donor oocyte conception and pregnancy complications: a systematic review and meta-analysis. BJOG : an international journal of obstetrics and gynaecology. 2016;123(9):1471–80. https://doi.org/10.1111/1471-0528.13910.
Blazquez A, Garcia D, Rodriguez A, Vassena R, Figueras F, Vernaeve V. Is oocyte donation a risk factor for preeclampsia? A systematic review and meta-analysis. J Assist Reprod Genet. 2016;33(7):855–63. https://doi.org/10.1007/s10815-016-0701-9.
Storgaard M, Loft A, Bergh C, Wennerholm UB, Soderstrom-Anttila V, Romundstad LB, et al. Obstetric and neonatal complications in pregnancies conceived after oocyte donation: a systematic review and meta-analysis. BJOG. 2017;124(4):561–72. https://doi.org/10.1111/1471-0528.14257.
Adams DH, Clark RA, Davies MJ, de LS. A meta-analysis of neonatal health outcomes from oocyte donation. J Dev Orig Health Dis. 2015:1–16. doi:S2040174415007898 [pii];https://doi.org/10.1017/S2040174415007898 [doi].
Mascarenhas M, Sunkara SK, Antonisamy B, Kamath MS. Higher risk of preterm birth and low birth weight following oocyte donation: a systematic review and meta-analysis. Eur J Obstet Gynecol Reprod Biol. 2017;218:60–7. https://doi.org/10.1016/j.ejogrb.2017.09.015.
Al Shammary M, Shaw A, Bacal V, Menzies-Toman D, Rozon C, Weir A, et al. Risk of lower birth weight and shorter gestation in oocyte donation pregnancies compared with other assisted reproductive technology methods: systematic review. J Obstet Gynaecol Can. 2019. https://doi.org/10.1016/j.jogc.2019.08.045.
Moreno-Sepulveda J, Checa MA. Risk of adverse perinatal outcomes after oocyte donation: a systematic review and meta-analysis. J Assist Reprod Genet. 2019;36(10):2017–37. https://doi.org/10.1007/s10815-019-01552-4.
Levron Y, Dviri M, Segol I, Yerushalmi GM, Hourvitz A, Orvieto R, et al. The “immunologic theory” of preeclampsia revisited: a lesson from donor oocyte gestations. Am J Obstet Gynecol. 2014;211(4):383.e1-5. https://doi.org/10.1016/j.ajog.2014.03.044.
Lutjen P, Trounson A, Leeton J, Findlay J, Wood C, Renou P. The establishment and maintenance of pregnancy using in vitro fertilization and embryo donation in a patient with primary ovarian failure. Nature. 1984;307(5947):174–5.
Ferraretti AP, Nygren K, Andersen AN, de Mouzon J, Kupka M, Calhaz-Jorge C et al. Trends over 15 years in ART in Europe: an analysis of 6 million cycles. Hum Reprod Open. 2017;2017(2):hox012. doi:https://doi.org/10.1093/hropen/hox012.
Van Bentem K, Lashley EELO, Van der hoorn MLP. The significance of specialized preconception counselling in oocyte donation pregnancy with prior history of postpartum eclampsia. BMJ Case Reports: accepted for publication. 2020.
Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6(7): e1000097. https://doi.org/10.1371/journal.pmed.1000097.
Berntsen S, Larsen EC, la Cour FN, Pinborg A. Pregnancy outcomes following oocyte donation. Best Pract Res Clin Obstet Gynaecol. 2020. https://doi.org/10.1016/j.bpobgyn.2020.07.008.
Rolnik DL, Wright D, Poon LC, O’Gorman N, Syngelaki A, de Paco MC, et al. Aspirin versus placebo in pregnancies at high risk for preterm preeclampsia. N Engl J Med. 2017;377(7):613–22. https://doi.org/10.1056/NEJMoa1704559.
Weckstein LN, Jacobson A, Galen D, Hampton K, Hammel J. Low-dose aspirin for oocyte donation recipients with a thin endometrium: prospective, randomized study. Fertil Steril. 1997;68(5):927–30.
Oocyte or embryo donation to women of advanced reproductive age: an Ethics Committee opinion. Fertil Steril. 2016;106(5):e3-e7. doi:https://doi.org/10.1016/j.fertnstert.2016.07.002.
Kamath MS, Mascarenhas M, Kirubakaran R, Bhattacharya S. Number of embryos for transfer following in vitro fertilisation or intra-cytoplasmic sperm injection. Cochrane Database Syst Rev. 2020;8:Cd003416. doi:https://doi.org/10.1002/14651858.CD003416.pub5.
Boria F, de la Calle M, Cuerva M, Sainz A, Bartha JL. Impact of oocyte donation on obstetric and perinatal complications in twin pregnancies. J Matern Fetal Neonatal Med. 2018:1–91. doi:https://doi.org/10.1080/14767058.2018.1533944.
Guilbaud L, Santulli P, Studer E, Gayet V, Goffinet F, Le Ray C. Impact of oocyte donation on perinatal outcome in twin pregnancies. Fertil Steril. 2017;107(4):948-53.e1. https://doi.org/10.1016/j.fertnstert.2017.01.019.
Webber L, Davies M, Anderson R, Bartlett J, Braat D, Cartwright B, et al. ESHRE guideline: management of women with premature ovarian insufficiency. Hum Reprod. 2016;31(5):926–37. https://doi.org/10.1093/humrep/dew027.
Geisler M, Meaney S, O’Donoghue K, Waterstone J. Oocyte donation pregnancies- non-disclosure of oocyte recipient status to obstetric care providers and perinatal outcomes. Ir Med J. 2017;110(10):654.
Mukhopadhaya N, Arulkumaran S. Reproductive outcomes after in-vitro fertilization. Curr Opin Obstet Gynecol. 2007;19(2):113–9. https://doi.org/10.1097/GCO.0b013e32807fb199.
Bodri D, Guillén JJ, Polo A, Trullenque M, Esteve C, Coll O. Complications related to ovarian stimulation and oocyte retrieval in 4052 oocyte donor cycles. Reprod Biomed Online. 2008;17(2):237–43. https://doi.org/10.1016/s1472-6483(10)60200-3.
Bennett SJ, Waterstone JJ, Cheng WC, Parsons J. Complications of transvaginal ultrasound-directed follicle aspiration: a review of 2670 consecutive procedures. J Assist Reprod Genet. 1993;10(1):72–7. https://doi.org/10.1007/bf01204444.
Dicker D, Ashkenazi J, Feldberg D, Levy T, Dekel A, Ben-Rafael Z. Severe abdominal complications after transvaginal ultrasonographically guided retrieval of oocytes for in vitro fertilization and embryo transfer. Fertil Steril. 1993;59(6):1313–5. https://doi.org/10.1016/s0015-0282(16)55997-4.
Roest J, Mous HV, Zeilmaker GH, Verhoeff A. The incidence of major clinical complications in a Dutch transport IVF programme. Hum Reprod Update. 1996;2(4):345–53. https://doi.org/10.1093/humupd/2.4.345.
Bustillo M, Buster JE, Cohen SW, Thorneycroft IH, Simon JA, Boyers SP, et al. Nonsurgical ovum transfer as a treatment in infertile women. Preliminary experience Jama. 1984;251(9):1171–3.
Antinori S, Versaci C, Gholami GH, Panci C, Caffa B. Oocyte donation in menopausal women. Hum Reprod. 1993;8(9):1487–90. https://doi.org/10.1093/oxfordjournals.humrep.a138284.
Klein J, Sauer MV. Oocyte donation. Best Pract Res Clin Obstet Gynaecol. 2002;16(3):277–91. https://doi.org/10.1053/beog.2002.0288.
Pados G, Camus M, Van Steirteghem A, Bonduelle M, Devroey P. The evolution and outcome of pregnancies from oocyte donation. Hum Reprod. 1994;9(3):538–42. https://doi.org/10.1093/oxfordjournals.humrep.a138541.
Sauer MV. Reproduction at an advanced maternal age and maternal health. Fertil Steril. 2015;103(5):1136–43. https://doi.org/10.1016/j.fertnstert.2015.03.004.
du Fosse NA, van der Hoorn MP, van Lith JMM, le Cessie S, Lashley E. Advanced paternal age is associated with an increased risk of spontaneous miscarriage: a systematic review and meta-analysis. Hum Reprod Update. 2020. https://doi.org/10.1093/humupd/dmaa010.
Oldereid NB, Wennerholm UB, Pinborg A, Loft A, Laivuori H, Petzold M, et al. The effect of paternal factors on perinatal and paediatric outcomes: a systematic review and meta-analysis. Hum Reprod Update. 2018;24(3):320–89. https://doi.org/10.1093/humupd/dmy005.
Dviri M, Madjunkova S, Koziarz A, Madjunkov M, Mashiach J, Nekolaichuk E, et al. Is there an association between paternal age and aneuploidy? Evidence from young donor oocyte-derived embryos: a systematic review and individual patient data meta-analysis. Hum Reprod Update. 2020. https://doi.org/10.1093/humupd/dmaa052.
Singh B, Reschke L, Segars J, Baker VL. Frozen-thawed embryo transfer: the potential importance of the corpus luteum in preventing obstetrical complications. Fertil Steril. 2020;113(2):252–7. https://doi.org/10.1016/j.fertnstert.2019.12.007.
Groenewoud ER, Cohlen BJ, Macklon NS. Programming the endometrium for deferred transfer of cryopreserved embryos: hormone replacement versus modified natural cycles. Fertil Steril. 2018;109(5):768–74. https://doi.org/10.1016/j.fertnstert.2018.02.135.
Sha T, Yin X, Cheng W, Massey IY. Pregnancy-related complications and perinatal outcomes resulting from transfer of cryopreserved versus fresh embryos in vitro fertilization: a meta-analysis. Fertil Steril. 2018;109(2):330-42.e9. https://doi.org/10.1016/j.fertnstert.2017.10.019.
Maheshwari A, Pandey S, Amalraj Raja E, Shetty A, Hamilton M, Bhattacharya S. Is frozen embryo transfer better for mothers and babies? Can cumulative meta-analysis provide a definitive answer? Hum Reprod Update. 2018;24(1):35–58. https://doi.org/10.1093/humupd/dmx031.
Sites CK, Wilson D, Barsky M, Bernson D, Bernstein IM, Boulet S, et al. Embryo cryopreservation and preeclampsia risk. Fertil Steril. 2017;108(5):784–90. https://doi.org/10.1016/j.fertnstert.2017.08.035.
Acknowledgements
The authors want to thank librarian J.W. Schoones (Walaeus Library, Leiden University Medical Center) for help with the literature search.
Author information
Authors and Affiliations
Contributions
M.L. van der Hoorn came up with the idea for the article. M.L. van der Hoorn and K. van Bentem performed the literature search, data analysis, and drafted the work. All authors critically revised the work.
Corresponding author
Ethics declarations
Ethics Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
van der Hoorn, ML., van Bentem, K. & Lashley, E. Evidence-Based Pre-Pregnancy Counseling for Oocyte Donation Pregnancies: a Systematic Review and Guide for Physicians. Reprod. Sci. 29, 3311–3320 (2022). https://doi.org/10.1007/s43032-021-00821-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-021-00821-x