
Dioxygenyl
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Dioxygenyl?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
For other uses of "O2", see O2 (disambiguation).
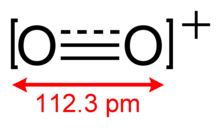
The dioxygenyl ion, O+
2, is a rarely-encountered oxycation in which both oxygen atoms have a formal oxidation state of +1/2. It is formally derived from oxygen by the removal of an electron:
- O2 → O+
2 + e−
Quick Facts Names, Identifiers ...
 | |
| Names | |
|---|---|
| IUPAC name
Dioxygenyl | |
| Identifiers | |
| ChEBI | |
| ChemSpider | |
| |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
The energy change for this process is called the ionization energy of the oxygen molecule. Relative to most molecules, this ionization energy is very high at 1175 kJ/mol.[1] As a result, the scope of the chemistry of O+
2 is quite limited, acting mainly as a 1-electron oxidiser.[2]